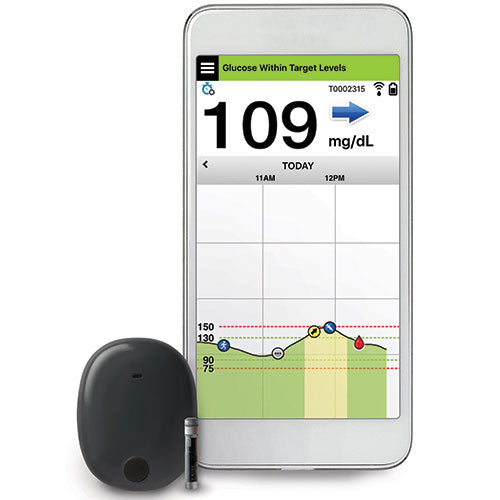
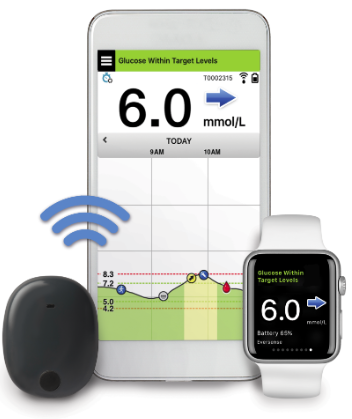
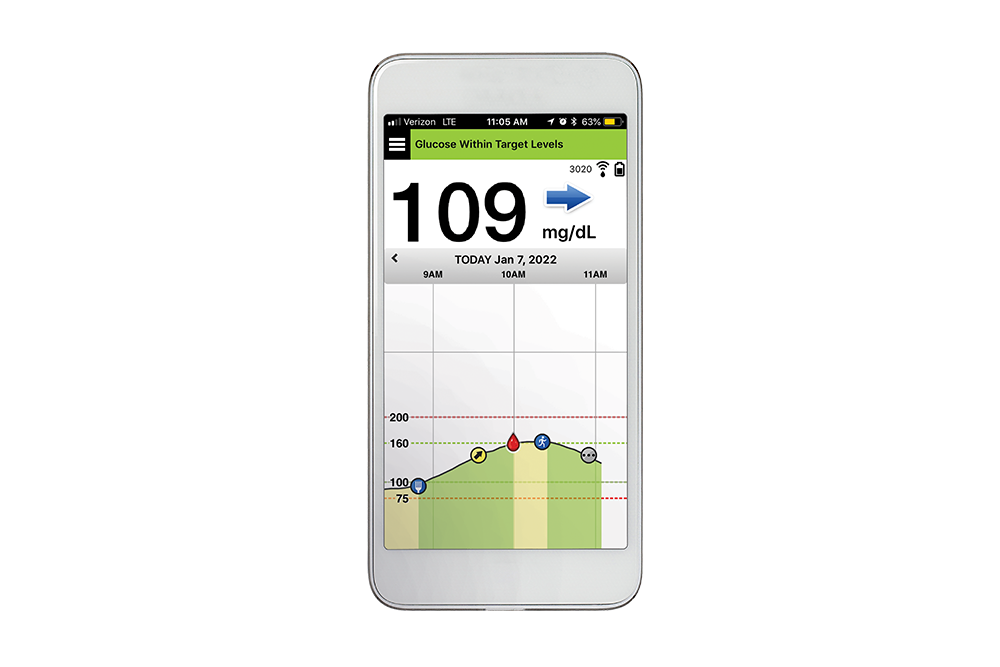
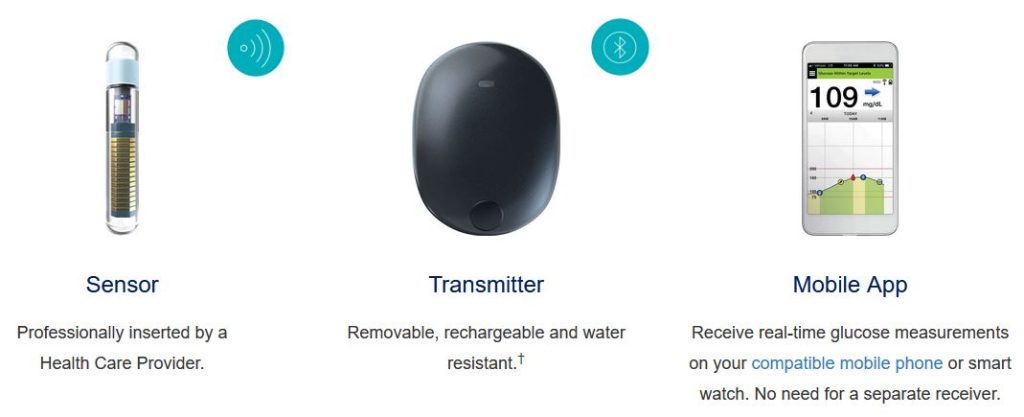
Ascensia Diabetes Care and Senseonics Announce 'The CGM for Real Life' Campaign to Raise Awareness of How Long-Term Eversense E3 Empowers People with Diabetes | Business Wire

Ascensia Diabetes Care and Senseonics Announce 'The CGM for Real Life' Campaign to Raise Awareness of How Long-Term Eversense E3 Empowers People with Diabetes